Tomorrow's Cell
Therapies, Today®
Pluristyx provides induced pluripotent stem cells and the expertise to accelerate the development and commercialization of revolutionary cellular therapies.
We Simplify the Stem Cell Therapy
Development Process

We Simplify the Stem Cell Therapy
Development Process
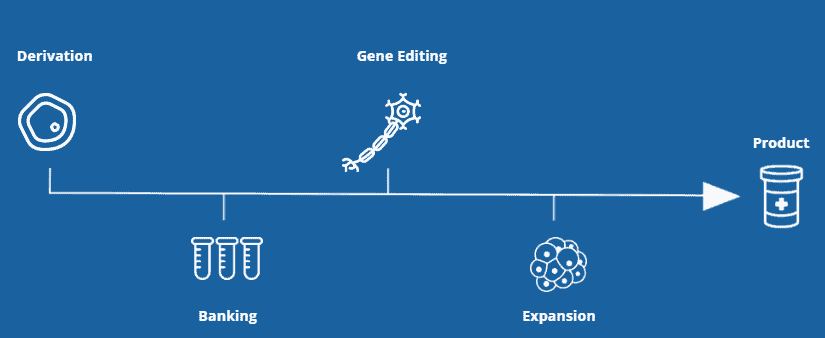

Why Pluristyx?
Pluristyx's innovative technologies, provides clients with the necessary expertise and tools to support and accelerate the development and commercialization of cellular products.
Our iPSC platform offers our clients a single point of access to our research and clinical grade stem cells, proprietary gene edits and end-to-end support throughout the product development pathway.
End-to-End Solutions
With our enlarged portfolio of unique non-modified and genetically engineered iPSC-based products, Pluristyx can assist you at any stage in your product development workflow.
Gene Edits
Offering an array of gene editing technologies and our proprietary FailSafe® edit resulting in unparalleled functional control of commercial products.
Clinical Path iPSC
Leading provider of gene-edited iPSC and cell therapy solutions, accelerating the path to clinic and providing the best route to commercialization.
Scientific Expertise
Veteran expertise from industry leaders in the field of stem cell biology, developmental genetics and cell therapy.
End-to-End Solutions
With our enlarged portfolio of unique non-modified and genetically engineered iPSC-based products, Pluristyx can assist you at any stage in your product development workflow.
Gene Edits
Offering an array of gene editing technologies and our proprietary FailSafe® edit resulting in unparalleled functional control of commercial products.
Clinical Path iPSC
Leading provider of gene-edited iPSC and cell therapy solutions, accelerating the path to clinic and providing the best route to commercialization.
Scientific Expertise
Veteran expertise from industry leaders in the field of stem cell biology, developmental genetics and cell therapy.
Try Before You Buy
Would you like to experience our technology firsthand before committing to a license agreement? We welcome you to have access to our various product offerings before you purchase a commercial license.
Your Problems, Our Solution
To overcome any obstacle you’re facing, Pluristyx offers a novel iPSC platform for rapid, efficacious, and high-quality iPS cell therapy development, can turn your bold ideas into reality.
Your Problems,
Our Solution
To overcome any obstacle you’re facing, Pluristyx offers a novel platform for rapid, efficacious, and high-quality iPSC therapy development, can turn your bold ideas into reality.

About Pluristyx
Pluristyx offers an enlarged portfolio of unique and effective non-modified and genetically engineered iPSC-based technologies and related services to provide end-to-end client support throughout the product lifecycle.

About Pluristyx
Pluristyx offers an enlarged portfolio of unique and effective non-modified and genetically engineered iPSC-based technologies and related services to provide end-to-end client support throughout the product lifecycle.
About Pluristyx

Pluristyx offers an enlarged portfolio of unique and effective non-modified and genetically engineered iPSC-based technologies and related services to provide end-to-end client support throughout the product lifecycle.
Latest News

Made Scientific and Pluristyx Launch Integrated iPSC Development & Manufacturing Partnership to Advance Next-Generation iPSC-derived Cell Therapies
Strategic partnership integrates Pluristyx's clinical-grade iPSC platform into Made Scientific's CDMO services, giving developers a single-source path from cell line to commercial supply. PRINCETON, NJ and Seattle WA, June 4, 2026 — Made Scientific, Inc., a leading U.S.-based clinical- and...

CellVoyant and Pluristyx Announce Strategic Partnership to Integrate Predictive AI with iPSC Technologies for Enhanced Cell Therapy Development
BRISTOL, UK and SEATTLE, WA, USA — 7 MAY 2026 — CellVoyant, a pioneer in AI foundation models for live, label-free cell analytics and predictive cell biology, and Pluristyx, a leading provider of induced pluripotent stem cell (iPSC) products and...
Pluristyx and Qkine Announce a Reciprocal Distribution Agreement in Support of Optimized Growth Factor Differentiation of Induced Pluripotent Stem Cell (iPSC)
SEATTLE, WA and CAMBRIDGE, UK — Pluristyx, a leading provider of clinical-grade induced pluripotent stem cell (iPSC) solutions, and Qkine, a pioneer in the manufacture of high-purity, animal-free growth factors and cytokines, today announced a distribution agreement to support streamlined...
