Price
Description
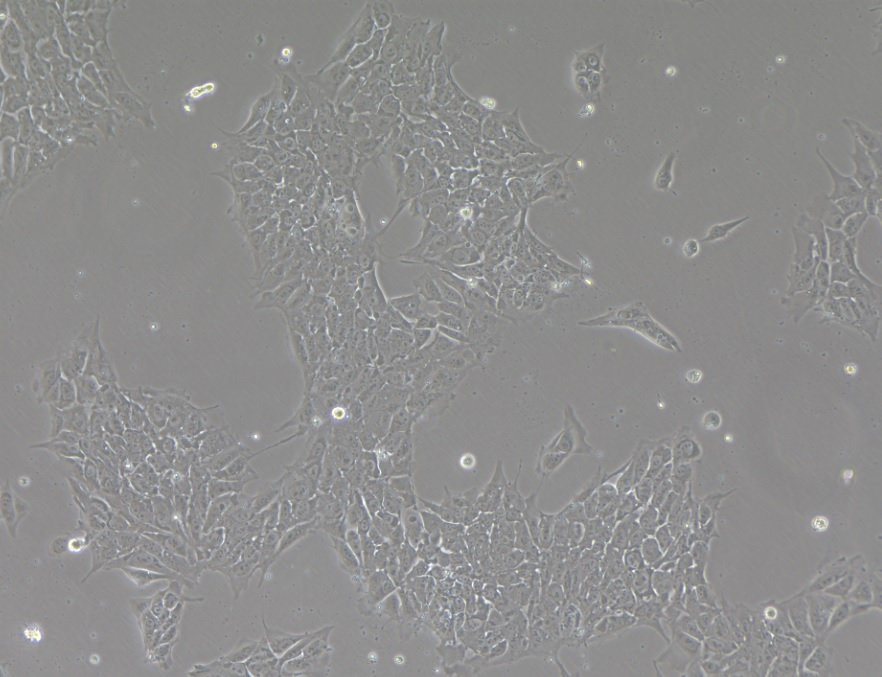
PLX 1.0 Human induced Pluripotent Stem Cells
Catalog Number: PLX04 001 R01
Cell number per vial: Please contact us for more information on the PLX 1.0 iPSC
Product Summary
PLX 1.0 Human Induced Pluripotent Stem Cells (hiPSCs) were derived from the dermis of juvenile foreskin procured under current Good Tissue Practice (cGTP) and conform to Global Ethical Standards and Clinical Cell Regulations. PLX 1.0 hiPSCs are intended for Research Use Only (RUO) and shall not be developed for diagnostic or therapeutic use. Pluristyx PLX 1.0 cells are free of mycoplasma, pathogens, bacteria, yeast, and fungus, and guaranteed to achieve >80% viability post-thaw at the indicated cell number. PLX 1.0 cells were characterized for karyotype, pluripotency, and Short Tandem Repeat (STR) cell line identity.
PLX 1.0 are available in different formats: standard bank vial (> 0.5 M cells), and Ready to Differentiate (RTD™) high density vials of pluripotent stem cells at 25 or 100 million cells/vial suitable for thaw and immediate use.
Format: Research Use Only; not for diagnostic or therapeutic purposes
Passage number: <25
Karyotype: 46, XY
Pluripotency Markers: OCT4 & SSEA-4 (>90%); SSEA-1 (<10%)
These cells are for Research Use Only (RUO) and shall not be developed for diagnostic, therapeutic, or commercial use. Please enquire with Pluristyx for GMP grade vials that may be used for clinical or commercial use.